3
THERAPEUTIC AREAS
7+
CLINICAL PARTNERS
100%
CANADIAN
Patient
Track your health journey and stay connected with your care team between visits.
Healthcare Professional
Make faster, evidence-based clinical decisions with continuous patient insights.
Explore ›Healthcare Organization
Scale precision care and drive operational efficiency across your institution.
Explore ›Research & Pharma
Accelerate evidence generation with structured real-world longitudinal data.
Explore ›Partnerships
Unlock scalable value through real-world clinical intelligence.
Explore ›
Three Therapeutic Areas,
Where Precision Monitoring Matters Most
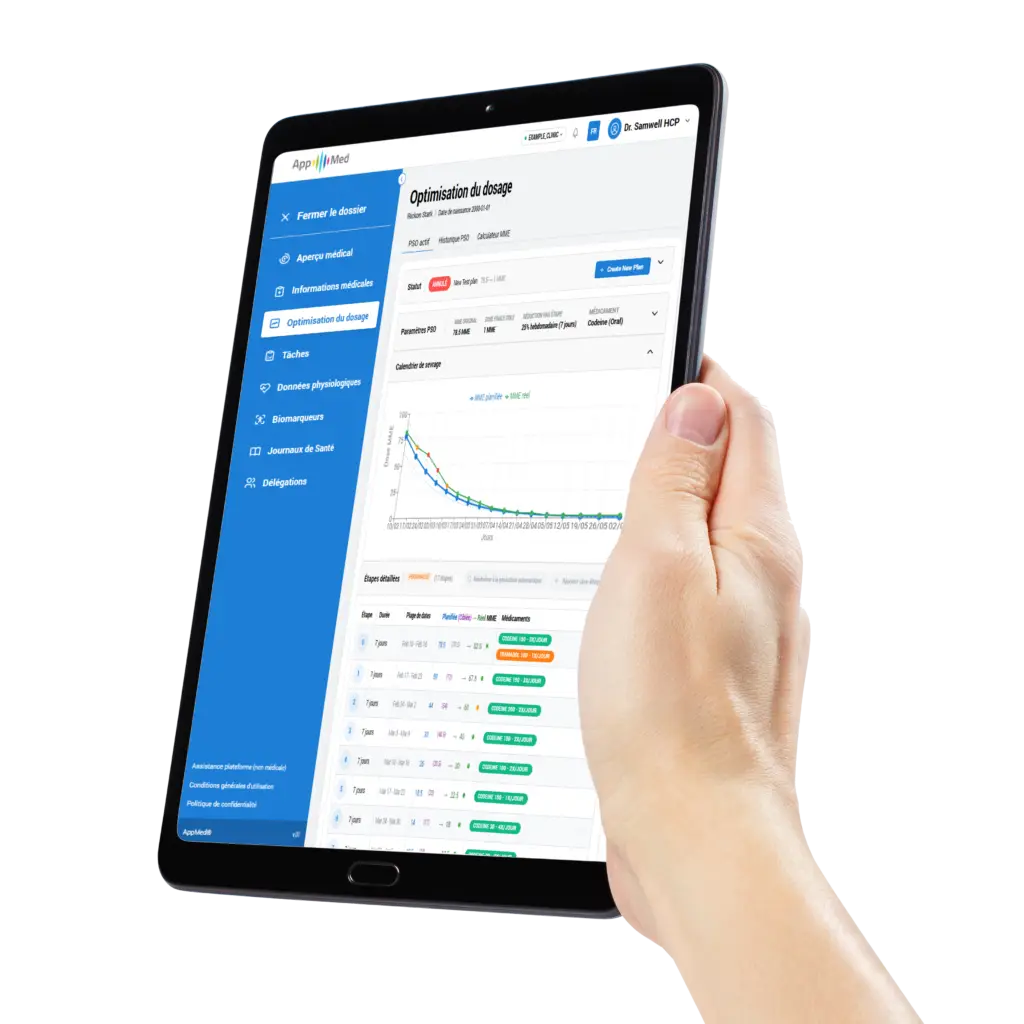
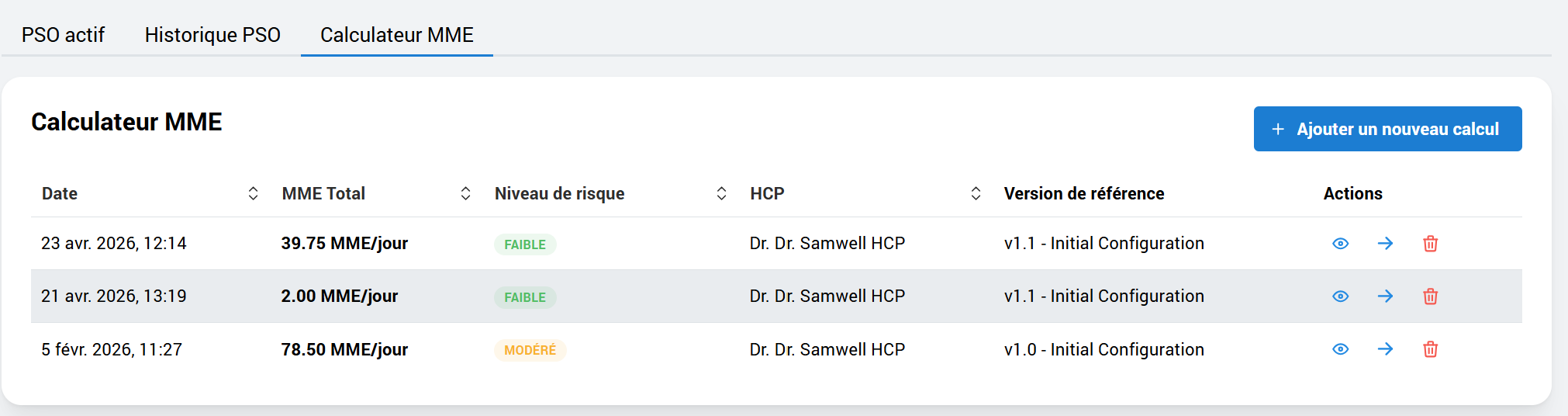
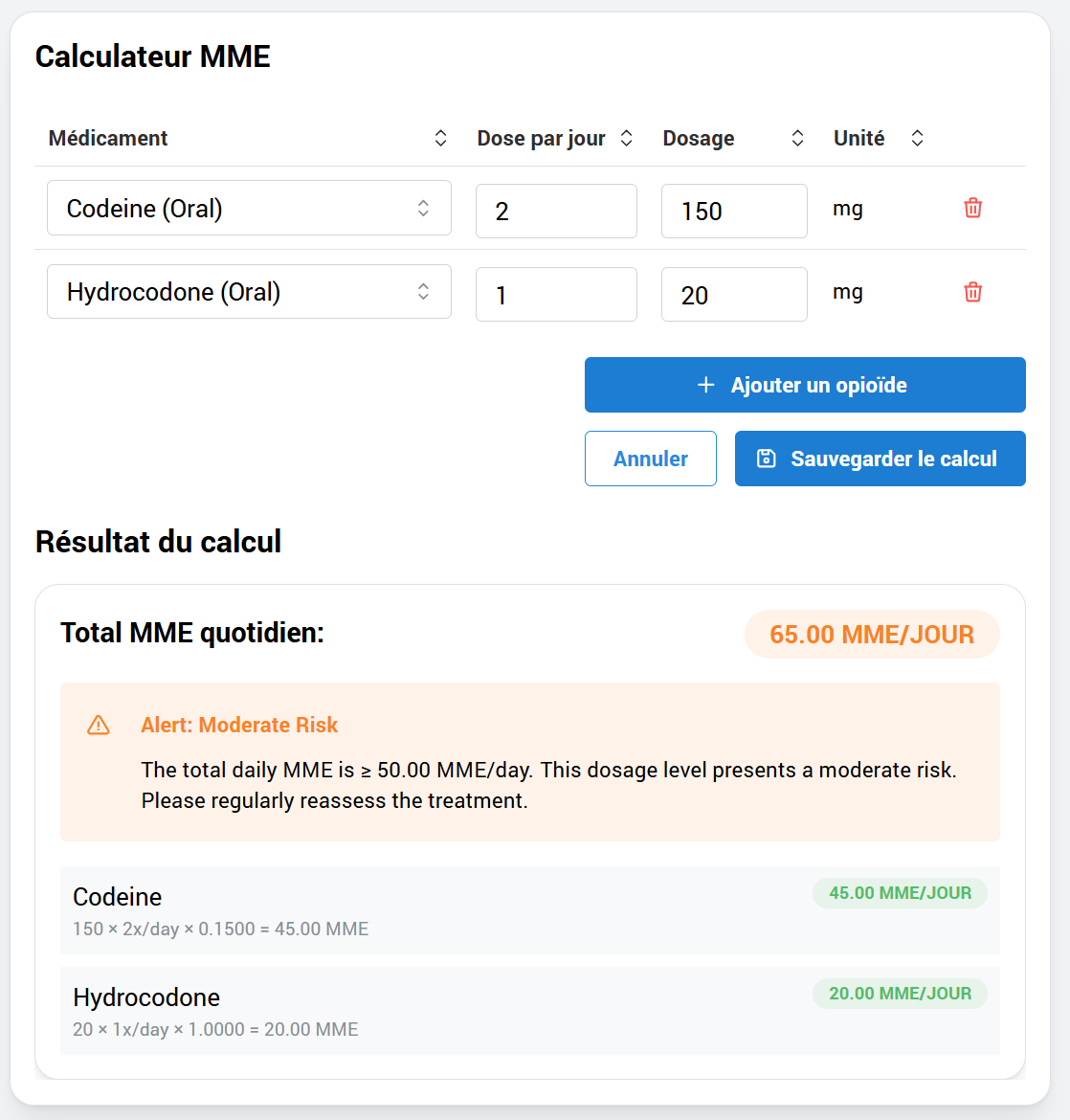
Post-operative pain management and deprescribing protocols, with an adherence tracking system and continuous pain assessments.
Menopause care with continuous real-time symptoms and period tracking to guide individualized treatment decisions.
Monitoring of neurodivergent conditions using validated tools and a population PK/PD calculator for ADHD, enabling real-time therapeutic decisions.
Infrastructure designed not just for uptime, but for clinical trust, regulatory alignment, and uninterrupted patient oversight.
Not just compliant, AppMed™ is built to operate within the highest standards of clinical data governance in Canada.
Not just encrypted, AppMed™ ensures that the right data is accessible only to the right people, at the right time, under full institutional control.
Not just certified infrastructure, AppMed™ delivers governed, accountable, and audit-ready security at the application level.