Real-World Evidence,
Structured at Scale
AppMed™ provides pharmaceutical companies and research institutions with a regulatory-grade infrastructure for generating longitudinal, multi-modal real-world evidence, directly from clinical care environments.
- PROs, biomarkers & adherence data — structured & longitudina
- Health Canada SaMD · Regulatory-grade data governance
- Ethics-ready consent frameworks built into every workflow
- Faster evidence generation than traditional clinical collection

3
Valided THERAPEUTIC AREAS
<4wk
deployment time
3+
Active Clinical Sites
100%
regulatory-grade data
paltform capability
Multi-Modal Data Capture, Structured Into
One Research-Ready Infrastructure
AppMed™ captures multi-modal patient data continuously and structures it for research and regulatory use, eliminating manual collection overhead and data quality issues.
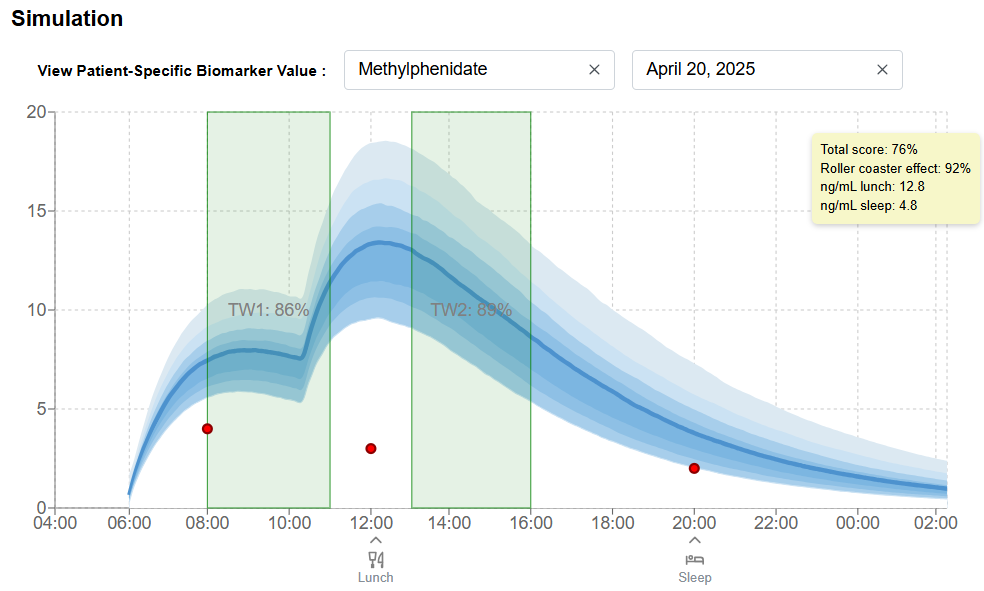
Model-Informed Precision Dosing
Validated pharmacokinetic and pharmacodynamic (PK/PD) models applied to individual patient data, generating dosing insight that can be used for both clinical practice and drug development programs. This function is ideal for medication with narrow therapeutic window (NTW).
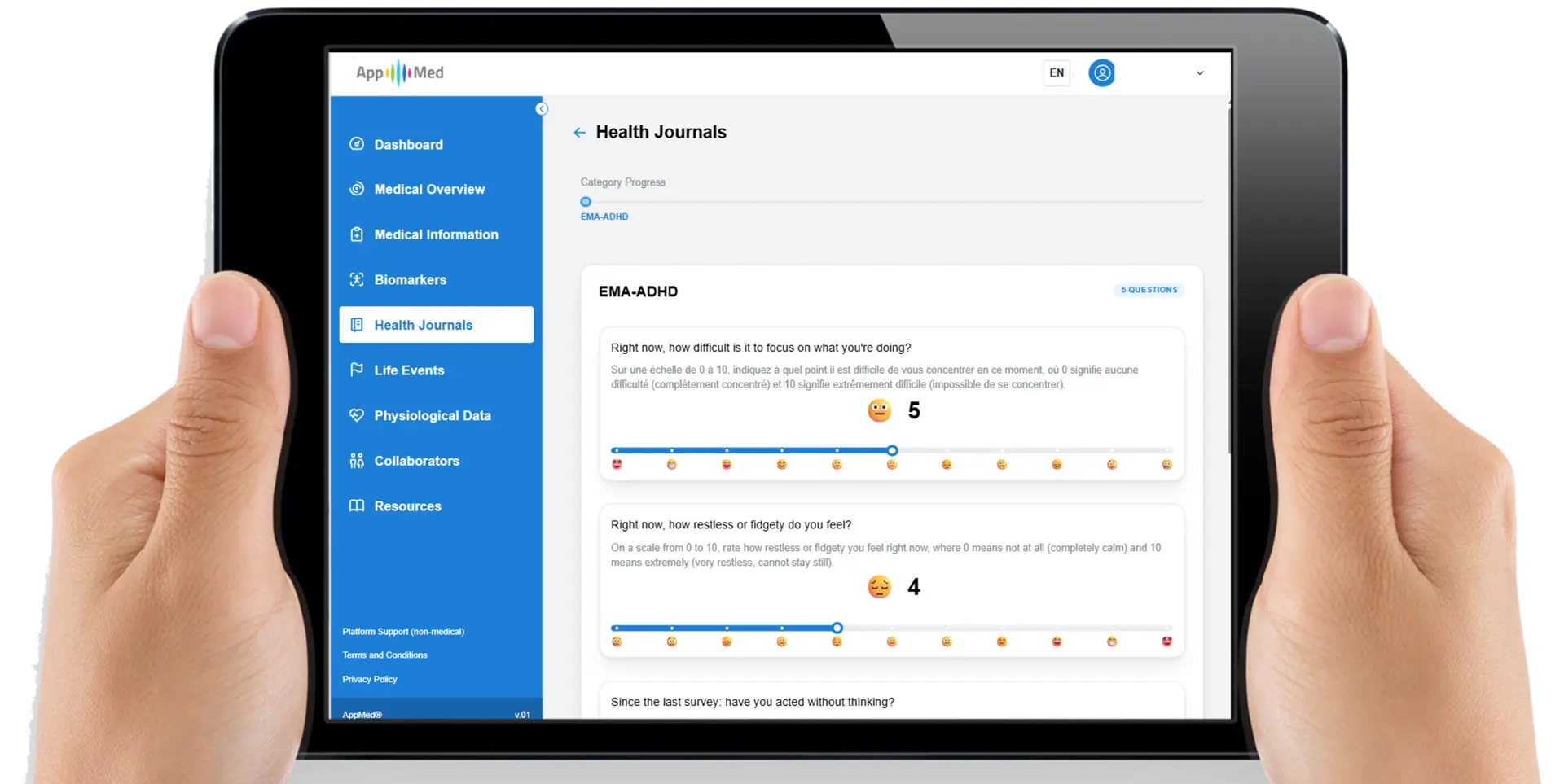
Patient-Reported Outcomes (PROs)
Validated, condition-specific questionnaires delivered continuously between clinical encounters — generating structured longitudinal symptom and quality-of-life data across enrolled populations.

Longitudinal Analytics & Export
Structured, analysis-ready datasets with full temporal resolution, exportable in research-grade formats for biostatistical analysis, regulatory submission support, or publication workflows.

Multi-Site & Federated Research
Deploy research protocols across multiple clinical sites simultaneously, with centralized data governance, standardized collection instruments, and unified analytics dashboards.

Adherence & Treatment Data
Objective medication adherence tracking via connected dispensing systems (MDS) providing real-world compliance data that is often missing from traditional clinical trial datasets.

Microsampling Biomarkers
Volumetric microsampling enables patients to self-collect blood sample at home, enabling assessment of objective PK/PD and Therapeutic Drug Monitoring (TDM) data without clinic visits. The convenience for patients can dramatically increasing sampling frequency and compliance.
The AppMed™ RWE Advantage
Evidence That Reflects Real Clinical Reality
Traditional clinical trial data often diverges from real-world patient behavior. AppMed™ captures what actually happens, between visits, at home, over time, giving you evidence that’s both scientifically rigorous and ecologically valid.
Continuous, Not Episodic
Data collected daily, not just at quarterly clinic visits, providing temporal resolution that traditional studies cannot achieve.
Objective, Not Recalled
Structured patient inputs replace unreliable recall, eliminating recall bias and improving data integrity across all endpoints.
Structured, Not Fragmented
Every data point is collected within a defined schema, ready for regulatory submission, publication, or licensing without manual cleaning.
Fast, Not Delayed
Evidence generation in months, not years. AppMed™'s existing clinical network gives you a deployed, consented population from day one.
Microsampling Technology
Objective Biomarker Data,
Collected at-Home
AppMed™ integrates volumetric microsampling, enabling patients to self-collect dried blood spot samples at home, which are then analyzed for pharmacokinetic and therapeutic drug monitoring endpoints.

Microsampling
Mitra® VAMS® Microsampling Technology
120µL
Max Collection
99%
Acceptance Rate
✓
Health Canada
No clinic visit required
for routine biomarker sampling, dramatically reducing patient burden and dropout.
Higher sampling frequency
than traditional venepuncture, enabling richer PK/PD modelling and time-course analysis.
Validated analytical methods
with laboratory-grade accuracy, defensible for regulatory submissions and publication.
Integrated into the AppMed™ workflow
sample logistics, results ingestion, and data structuring handled automatically.
VAMS® Technology · Blood Microsampling
Easily Collect Up to 120µL of Blood and Ship by Mail to Laboratory
The Mitra® device was developed with Volumetric Absorptive Microsampling (VAMS®) for easy remote blood collection by virtually anyone, anywhere, anytime — offering a streamlined, flexible, and decentralized approach to biological specimen collection.
- Easier trial recruitment and increased patient adherence to sampling protocols
- Elimination of cold-chain shipping and storage requirements
- VAMS® technology eliminates hematocrit bias for consistent quantitative results
- Outer housing protects against contamination during transit
- Reduced reliance on trained staff — patients self-collect at home
Class 1 IVD Health Canada Registered
How We Work Together
Flexible Research & Commercial
Partnership Model
Whether you need access to structured real-world data, a deployment platform for your study, or an integrated research collaboration, AppMed™ has a model that fits.
Data Licensing
Structured Real-World Data Access
License structured, longitudinal, de-identified datasets from AppMed™’s clinical network, across therapeutic areas, patient cohorts, and data modalities, for post-market research and population analytics.
Study Infrastructure
Deploy Your Research Protocol on AppMed™
Use AppMed™ as the digital clinical research platform for your investigator-initiated or sponsored study, leveraging our existing patient population, consent framework, and data collection infrastructure.
Pharma Collaboration
Adherence & RWE for Drug Programs
Integrate AppMed™ into your lifecycle management, post-approval, or label expansion programs, generating real-world adherence, efficacy, and safety data for regulatory and commercial purposes.
Academic Research
Investigator-Initiated Research Access
Research institutions and academic medical centres can access AppMed™ as a platform for investigator-initiated studies, with ethics-ready frameworks, structured data collection, and multi-site coordination support.
Regulatory & Data Governance
Data You Can Defend
In Any Regulatory Environment
AppMed™’s data infrastructure is built to the standards required for regulatory submission, ethics board review, and international publication, not just for internal use.
Health Canada SaMD Class II
AppMed™ is developed as a Software-as-a-Medical-Device aligned with Health Canada requirements, providing a regulatory-defensible foundation for data collected within the platform.
Full Audit Trail & Traceability
Every data point, access event, and modification is logged with timestamp and user attribution, supporting GCP-aligned audit requirements and regulatory inspection readiness.
Ethics-Ready Consent Framework
Granular, purpose-specific, and revocable consent is embedded into every patient workflow — enabling ethics board submission with a clear, auditable consent trail for each data use.
Loi 25 · PIPEDA · AWS Montréal
All data hosted exclusively in Québec-based AWS infrastructure, ensuring full Canadian data sovereignty, applicable to Loi 25 and PIPEDA, with no international data transfer by default.
Start a Research Partnership
Generate Evidence That Moves
Drug Research Forward
Whether you need structured real-world data, a research platform, or a pharma collaboration, let’s explore what’s possible with AppMed™’s clinical infrastructure.
Responses within 48 hours · NDA available on request · Quebec-based team
